Category: Medications
-
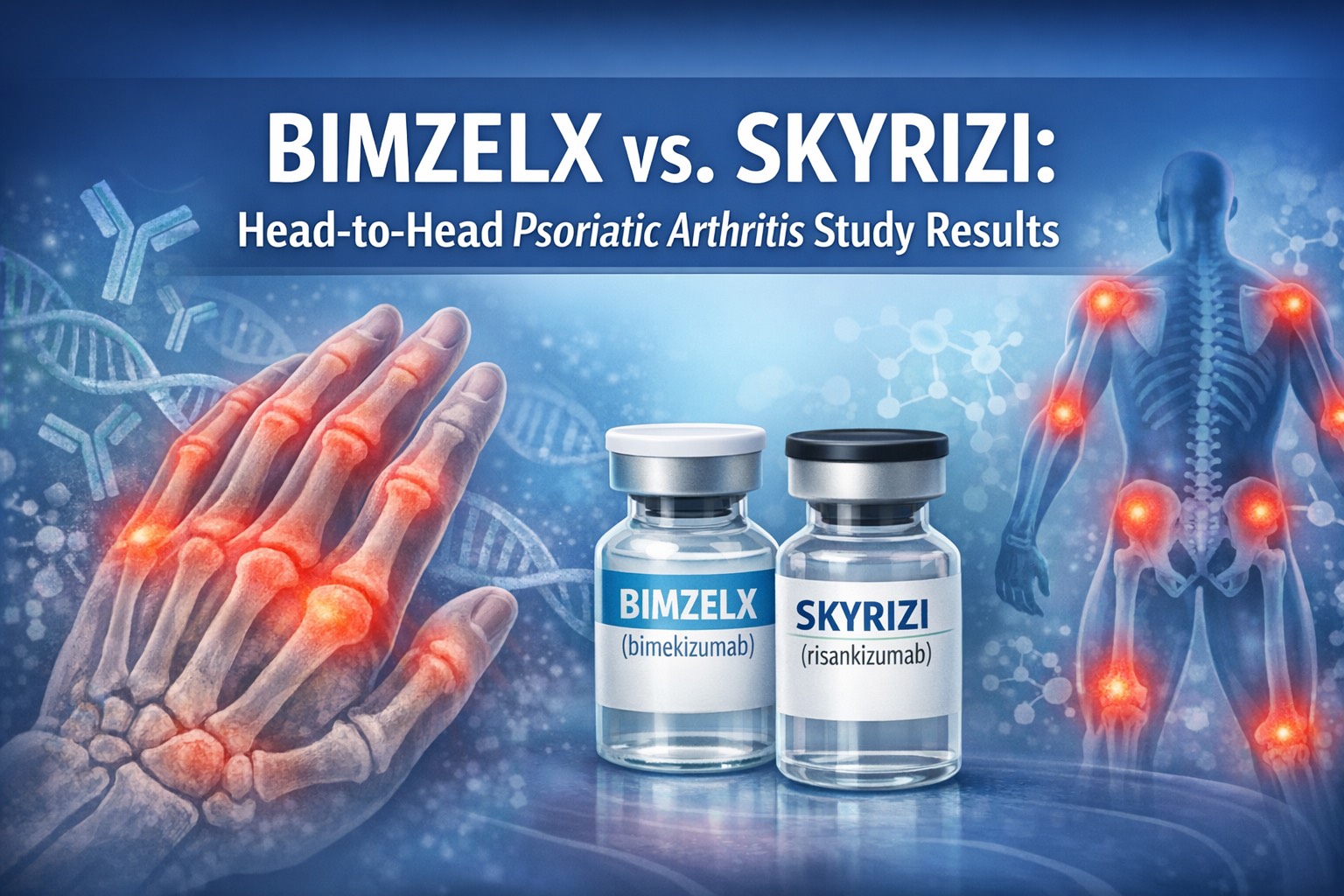
BIMZELX vs SKYRIZI – one stands out in a head‑to‑head trial
A new head‑to‑head study found that bimekizumab (brand name BIMZELX) produced better short‑term joint improvement than risankizumab (brand name SKYRIZI) in adults with active psoriatic arthritis (PsA). The trial’s primary measure was ACR50 – a commonly used, fairly strict standard that means a patient has achieved at least a 50% improvement in joint signs and
-

Universal nasal vaccine for coughs, colds and flu. How a single spray could change winter.
Imagine a single nasal spray that cuts your chances of catching the flu, COVID, the common cold, and even some bacterial lung infections – and that also calms allergic reactions that trigger asthma. That’s the bold idea behind a new line of research showing a nasal vaccine can supercharge the lungs’ frontline defenses, protecting mice
-

FDA Approves Rybrevant Faspro: A Faster, Simpler Option for Certain Lung Cancer Patients
The U.S. Food and Drug Administration (FDA) has approved Rybrevant Faspro, a newer formulation of the cancer drug amivantamab, offering a more convenient way to treat certain people with advanced lung cancer. While the science behind the drug is complex, the impact on patients’ daily lives is easier to understand: shorter treatment visits, fewer clinic
-

Vybrique Now FDA Approved: A New, Discreet Oral Film for Erectile Dysfunction
Erectile dysfunction (ED) affects millions of men and can be stressful to manage. A new option just cleared by the U.S. Food and Drug Administration (FDA) may make treatment easier and more discreet: Vybrique™, the first FDA‑approved sildenafil oral film that dissolves on the tongue. What is Vybrique and why does it matter? Vybrique is
-

FDA Approves Darzalex Faspro as Part of a New First‑Line Quadruplet Regimen for Multiple Myeloma
Multiple myeloma is a type of blood cancer that affects plasma cells – specialized white blood cells that help the body fight infections. For years, treatment options have steadily improved, but the newest development marks one of the most meaningful steps forward yet. The U.S. Food and Drug Administration (FDA) has approved a new four‑drug
-

Levalbuterol Tartrate Uses, Safety, and Alternatives
Levalbuterol tartrate is an effective short‑acting bronchodilator for acute bronchospasm and is an alternative to albuterol. For most patients, clinical outcomes are similar between levalbuterol and albuterol; cost, individual tolerance, and device preference usually determine which product is best. Always use rescue inhalers as directed, maintain controller therapy when prescribed, and seek medical care for