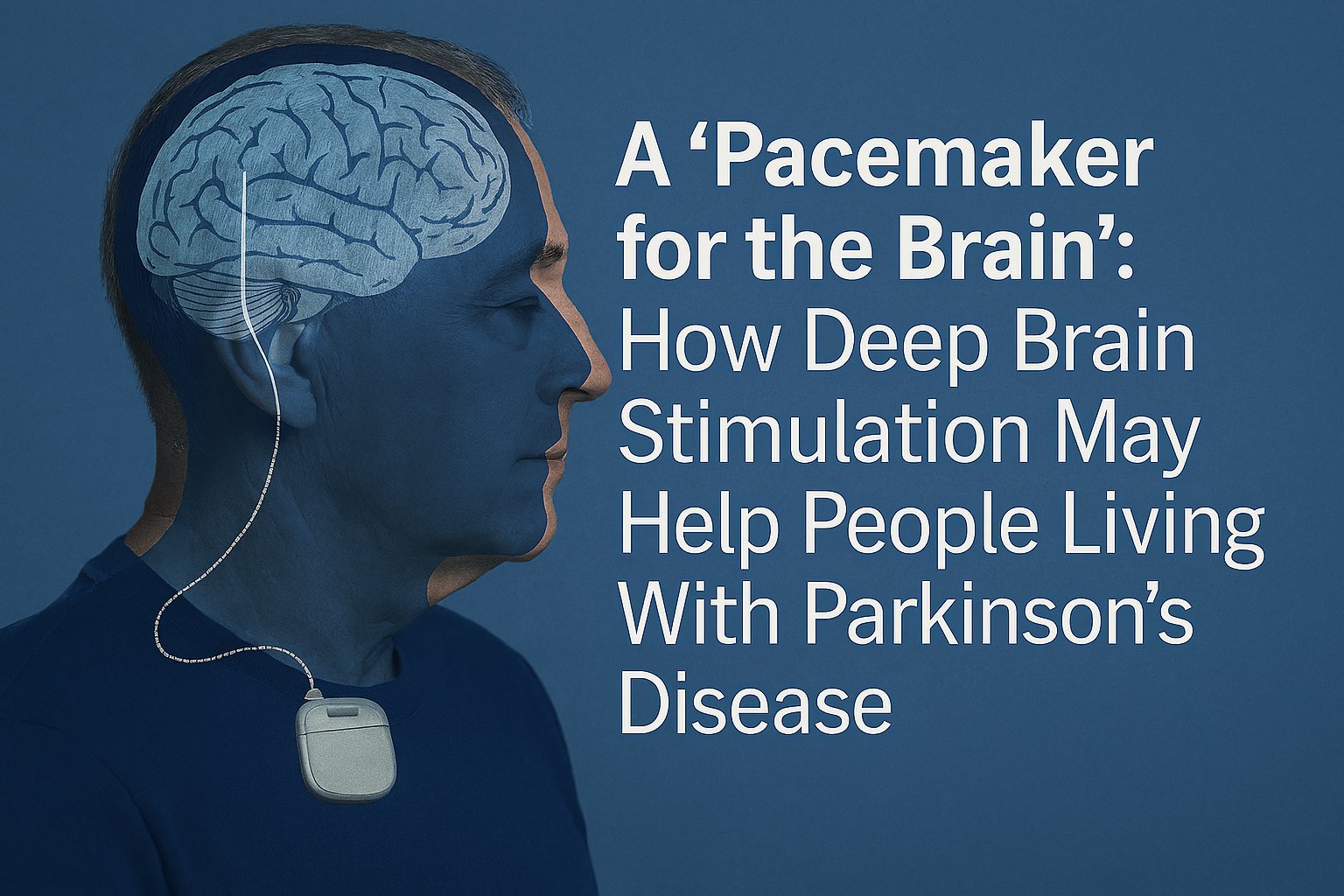
Deep brain stimulation – often described as a “pacemaker for the brain” – has become one of the most promising medical technologies for people with Parkinson’s disease whose symptoms are no longer well‑controlled by medication alone. While it doesn’t cure the disease, it can meaningfully reduce tremors, stiffness, and movement problems that interfere with daily life. Recent advances, including adaptive systems that automatically adjust stimulation in real time, are pushing this technology into an even more hopeful era.
What Parkinson’s Disease Does to the Brain
Parkinson’s disease affects a part of the brain called the substantia nigra, which normally produces dopamine – a chemical that helps coordinate smooth, controlled movement. As dopamine‑producing cells die off, the brain’s movement circuits begin to misfire. This leads to symptoms such as:
- Tremors
- Muscle stiffness
- Slowed movement
- Balance problems
Medications like levodopa can help, but over time they may become less effective or cause side effects such as involuntary movements. That’s where DBS may come in.
How Deep Brain Stimulation Works
DBS involves placing thin wires (electrodes) into specific areas of the brain that control movement. These wires connect to a small device implanted under the skin near the collarbone. The device sends controlled electrical pulses to help “reset” abnormal brain activity.
The Parkinson’s Foundation explains that DBS can reduce tremor, stiffness, slowness, and medication‑related fluctuations when drugs alone are no longer enough .
The system has three main parts:
- Leads: thin wires placed in targeted brain regions
- A neurostimulator: a battery‑powered device under the skin
- A controller: used by clinicians to adjust settings over time
This setup is why DBS is often compared to a pacemaker. Just as a heart pacemaker stabilizes abnormal heart rhythms, DBS stabilizes abnormal brain rhythms.
Why It’s Called a “Pacemaker for the Brain”
The comparison isn’t just a metaphor. Traditional pacemakers deliver steady electrical pulses to keep the heart beating properly. DBS does something similar for the brain’s movement circuits.
Stanford researchers describe DBS as providing electrical stimulation to “control the brain’s electrical rhythms,” much like a cardiac pacemaker controls heart rhythms .
But the newest generation of DBS devices goes even further.
The Rise of Adaptive Deep Brain Stimulation (aDBS)
Traditional DBS delivers constant stimulation, even when symptoms fluctuate throughout the day. But Parkinson’s symptoms – and the brain waves associated with them – aren’t constant.
Adaptive DBS (aDBS) changes that. It “listens” to the brain’s electrical activity and adjusts stimulation automatically. When abnormal brain waves rise, the device increases stimulation; when they fall, it eases off.
This makes the therapy more personalized and may reduce side effects. According to Stanford Medicine, aDBS corrects abnormal beta‑wave activity only when needed, making it more precise and efficient than older systems .
What the Latest Research Shows
A major international clinical trial published in JAMA Neurology in 2025 found that long‑term adaptive DBS was safe, effective, and well‑tolerated in people with Parkinson’s disease who were already using traditional DBS. Most participants experienced stable symptom control without troublesome side effects, and the adaptive system reduced the amount of electrical energy needed compared to continuous stimulation .
Key findings included:
- Most participants achieved good symptom control without increased dyskinesia.
- Adaptive systems reduce overall stimulation energy.
- No serious device‑related complications were reported during long‑term follow‑up.
These results suggest that adaptive DBS may offer more consistent symptom relief with fewer side effects.
What Symptoms DBS Helps Most
DBS is especially effective for symptoms that respond to levodopa, including:
- Tremor
- Stiffness
- Slowness
- Medication “on/off” fluctuations
It may also reduce the amount of medication needed, which can help minimize side effects.
However, DBS is less effective for:
- Speech problems
- Balance issues
- Cognitive symptoms
These symptoms often stem from different brain pathways that DBS does not target.
Who Might Be a Good Candidate?
DBS is typically considered when:
- Medications no longer control symptoms well
- Side effects from medication become difficult
- Tremor remains severe despite treatment
- Symptoms interfere with daily activities
Candidates usually undergo extensive evaluation, including neurological exams, imaging, and sometimes psychological testing. Because DBS involves brain surgery, the decision is made carefully with a medical team.
Risks and Considerations
Like any surgery, DBS carries risks. A systematic review published in BMJ Medicine found that DBS may increase the risk of serious adverse events, mainly due to surgical complications such as bleeding or infection, as well as hardware‑related issues like device malfunction .
Potential risks include:
- Infection at the implant site
- Bleeding in the brain
- Device complications requiring revision
- Temporary confusion after surgery
These risks are weighed against the potential benefits, especially for people whose symptoms are significantly affecting quality of life.
How DBS Is Evolving
DBS technology continues to advance. Researchers are exploring:
- More precise targeting using advanced imaging
- New brain regions that may help with tremor or gait problems
- Network‑based approaches that treat the brain as a connected system
- Smarter algorithms that adapt to real‑time brain activity
A 2025 narrative review in Biomedicines highlights how improved imaging and individualized targeting may lead to better outcomes and fewer side effects in the future .
What This Means for Patients and Families
For many people with Parkinson’s disease, DBS can provide meaningful improvements in daily life – helping them move more easily, reduce tremors, and regain independence. It’s not a cure, and it doesn’t stop the disease from progressing, but it can make symptoms more manageable for years.
A national study from UF Health also found that DBS can improve quality of life and that these benefits can last over time .
As adaptive systems become more widely available, DBS may become even more effective and personalized.
Better Treatment, Lower Cost – No Catch.
Find safer, more effective medications with fewer side effects – often for less money. It’s fast, free, and personalized. Learn More →
Sources (5)
- Parkinson’s Foundation – Deep Brain Stimulation (DBS) Overview
https://www.parkinson.org/living-with-parkinsons/treatment/surgical-treatment-options/deep-brain-stimulation - Stanford Medicine – Research Behind Adaptive Deep Brain Stimulation
https://med.stanford.edu/news/all-news/2025/02/deep-adaptive-brain-stimulation-parkinsons.html - JAMA Neurology – Long-Term Personalized Adaptive DBS Clinical Trial
https://jamanetwork.com/journals/jamaneurology/fullarticle/10.1001/jamaneurol.2025.2781 - Biomedicines – Deep Brain Stimulation for Parkinson’s Disease: Narrative Review
https://www.mdpi.com/2227-9059/13/10/2430 - UF Health – Study on Long-Term DBS Benefits
https://ufhealth.org/news/2025/uf-led-study-shows-deep-brain-stimulation-benefits-for-parkinsons-patients